Phytochemicals & Phytoconstituents
Under the label term "phytochemicals" falls a subset of nutritious plant chemicals, called phytonutrients-biologically effective plant materials that have been ascertained to be essential to maintain lives-in other words, vitamins and minerals discovered in herbs.
Unlike phytochemicals, Phytonutrients were ascertained to be effective to improve life.
We know how much consumption of phytonutrients is enough to prevent certain illnesses.
However, science has yet to determine the particular amounts of many phytochemicals necessarily to prevent disease.
Although there are hundreds of distinct phytochemicals, some of them are equal in function or composition and therefore, are classified in several divisions.
It is now established and fully believed that phytoconstituents obtained from the medicinal plants serve as pilot molecules in the modern medicines (Ncube et al., 2008) and many people still depend on the traditional medicine for their preliminary health care and treatment (Bannerman et al., 1986)
Following are some of those phytochemical families:
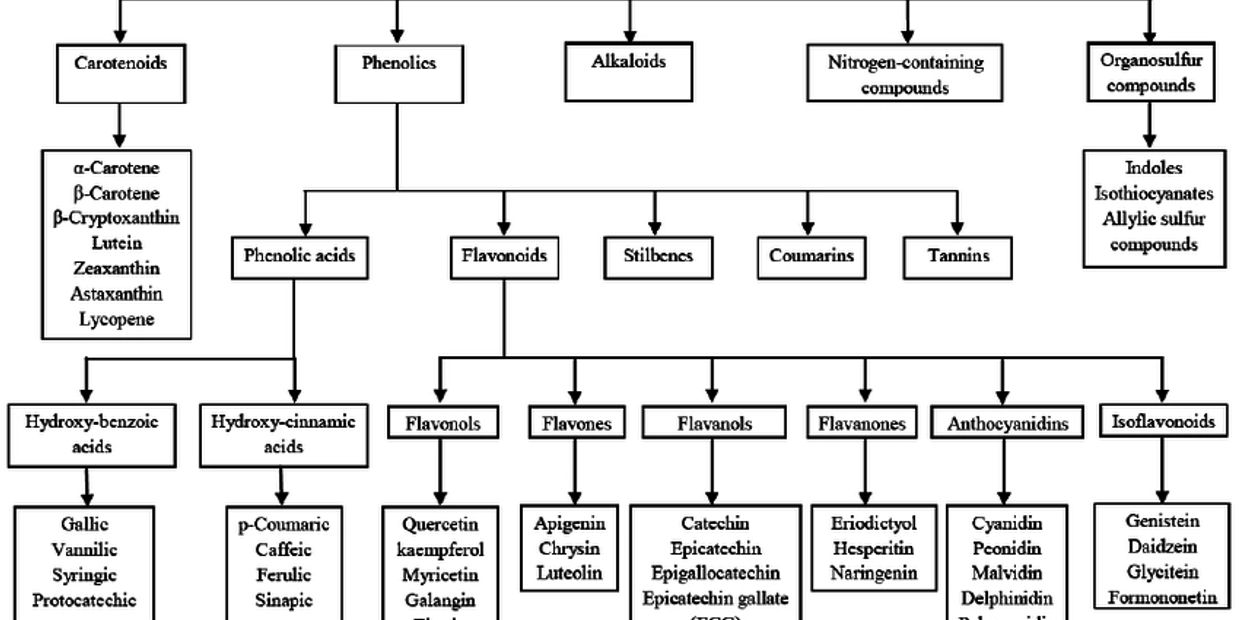
Carotenoids

The eye and skin boosters
Terpenes
Phytoconstituents
Phytoconstituents
Carotenes, limonoids, saponins
- Carotenoids such as beta-carotene are responsible for the orange-red colours of foods. Beta-carotene is converted in the body to vitamin A by a specific enzyme present in the gut.
Phytoconstituents
Phytoconstituents
Phytoconstituents
- Beta-Carotene
- Lycopene
- Lutein/Zeaxanthin
- Beta-Cryptoxanthin
Function
Function
Function
- Activate body's protective enzymes, protect eyes, act as antioxidants, modify hormones, help block cholesterol absorption, protect cellular differentiation
Foods
Function
Function
- Green, red and yellow vegetables and fruits; grains; legumes; nuts; seeds; herbs such as ginseng, chamomile, gotu kola, mango, carrots, tomatoes, sweet potato
broccoli, pumpkin, asparagus, peaches, kale, spinach
Isothiocyanates

Normal Cell Cycle Regulation
Organosulfur compounds
Organosulfur compounds
Organosulfur compounds
- Indol-3-carbinol,
- thiosulfonates,
- isothiocyanates
Phytoconstituents
Organosulfur compounds
Organosulfur compounds
1. Isothiocyanate
2. Sulforaphene
3. Phenethyl isothiocyanates
4. Allyl isothiocyanate
Function
Function
Function
- Boost cancer-fighting enzymes,
- block mutagenesis,
- inhibit cholesterol synthesis,
- may lower blood pressure
Food
Function
Function
Cruciferous vegetables; mustard family; onion family.
The vegetables that fall into this category include:
- kale,
- cauliflower,
- cabbage,
- broccoli, and collards, among others.
Phenols

Healthy-aging
Organusulfur compounds
Organusulfur compounds
Organusulfur compounds
- Polyphenols,
- anthocyanidins,
- catechins,
- isoflavones,
- tannins
Phytoconstituents
Organusulfur compounds
Organusulfur compounds
- flavonoids,
- terpenoids,
- tannins,
- xanthones
- phospholipids (phosphatidylcholine, phosphatidylserine)
Function
Function
Function
- Protect heart and vascular system,
- protect against colon cancer,
- modify hormone response,
- prevent dental caries
Food
Function
Function
- Berries,
- grapes,
- red wine,
- green leafy vegetables,
- soy foods,
- green tea, herbs
POlysaccharides

Manage viral infections
Organosulfur compounds
Organosulfur compounds
Organosulfur compounds
- Organic acids,
- Polysaccharides
Phytoconstituents
Organosulfur compounds
Organosulfur compounds
- Lactones,
- celluloses,
- arabinogalactans,
- pectins,
- fructans,
- glucans
Function
Function
Function
- Block nitrosamine effects,
- promote growth of beneficial intestinal bacteria,
- modulate immune system,
- may help prevent colon cancer
Food
Function
Function
- Fruit,
- mushrooms,
- yeast,
- herbs,
- spices
lipids

Manage inflammation
Organosulfur compounds
Organosulfur compounds
Organosulfur compounds
- Isoprenoids,
- oils,
- fatty acids,
- phytosterols
Phytoconstituents
Organosulfur compounds
Organosulfur compounds
- Phyto-phospholipid complex - Phytosomes
- Phospholipides
Function
Function
Function
- Reduce platelet aggregation,
- blood clotting,
- inflammation,
- nervous system disorders; balance hormones;
- modify autoimmune conditions
Food
Function
Function
- Dark-green leafy vegetables,
- nuts,
- soy oil,
- wheat germ,
- herbs,
- animal foods

Additional Information
Glycoside is one of a wide range of naturally occurring substances in which a component of starch composed of one or more sugars or uronic acid (i.e., a sugar acid) is mixed with a hydroxy compound.
The hydroxy compound, typically a non-sugar substance (aglycon), such as a phenol derivative or alcohol, may also be another carbohydrate, such as cellulose, glycogen, or starch, which comprises of several other units of glucose.
Many glycosides occur in plants, often as pigments for flowers and fruits, such as anthocyanins.
Various medicines, condiments, and plant dyes occur as glycosides; the heart stimulating glycosides of Digitalis and Strophanthus, members of a group known as cardiac glycosides, are of great value.
Glycosides (e.g., streptomycin) represent some antibiotics.
Glycosides derived from glucuronic acid and steroids are components of normal animal urine. Glycosides are also compounds (nucleosides) derived from partial nucleic acid degradation.
Formation of Glycosides
The hydroxyl group bound to the anomeric carbon atom (i.e., aldehyde-containing carbon or keto group) of carbohydrates in solution has peculiar reactivity and derivatives, called glycosides, can be formed; glucose-formed glycosides are called glucosides.
Balancing of a glycoside's α- and β-anomers in solution (i.e. mutarotation) can not occur.
The mechanism by which a glycoside is formed includes the anomeric carbon atom (numbered 1) hydroxyl group (−OH) of both α and β forms of d-glucose—α and β forms of d-glucose are shown in equilibrium in the reaction sequence — and the alcohol hydroxyl group (methyl alcohol in the reaction sequence); methyl α-d-glucosides and β-d-glucosides are formed as products, as is liquid.
Among the wide variety of naturally occurring glycosides are a number of plant pigments, particularly those colored red, violet, and blue; these pigments are found in flowers and consist of a pigment molecule, often glucose, attached to a sugar molecule.
In the preparation of indigo dye, plant indican (from Indigofera species), made up of glucose and indoxyl pigment, was essential before artificial dyes were prevalent. Digitalis is still being used of a variety of heart muscle stimulants that occur as glycosides.
Certain naturally occurring glycosides include vanillin present in the vanilla bean and amygdalin (bitter almond oil); a variety of glycosides contained in mustard has a sulfur atom in position 1 rather than oxygen.
Glycosides are a number of important antibiotics; streptomycin and erythromycin are among the best-known.
Glucosides, i.e. glycosides formed from glucose, are extremely important biological compounds in which the anomeric carbon atom is associated with phosphoric acid.
For example, in the biosynthesis of cellulose, starch, and glycogen, α-d-glucose-1-phosphate is an intermediate product; related glycosid phosphate derivatives of other monosaccharides are involved in the production of naturally occurring glycosides and polysaccharides.
A number of reactions can occur in hydroxyl groups other than the one at the anomeric carbon atom. Esterification, which consists of reacting with an appropriate acidic compound to the hydroxyl groups, results in the formation of a compound class called sugar esters.
The sugar acetates, in which the acid is acetic acid, are among the common ones.
Phosphoric acid esters and sulfuric acid are essential biological compounds; for example, glucose-6-phosphate plays a central role in most living cells ' energy metabolism, and d-ribulose 1,5-diphosphate plays an important role in photosynthesis.
Polyphenols and their involvement in nutrition
Polyphenols and Antioxidants
Polyphenols are phytochemicals, indicating the concentration of compounds found in natural plant food sources with antioxidant properties. In food products such as tea, coffee, chocolates, nuts, vegetables, and extra virgin olive oil, there are over 8,000 known polyphenols, just to mention a very few.
Polyphenols play a key role in preserving our health and well-being. As a group antioxidants help protect the body's cells from free radical destruction and regulate the speed at which you mature.
Free radicals can become rampant if our bodies does not get adequate protection, allowing your cells to perform poorly. This can result in tissue weakening which, for instance, put you at risk of conditions such as heart disease, leukemia, and Alzheimer's disease.
Antioxidants can be divided into three major groups:
- Allyl sulfides
- Carotenoids
- Polyphenols
Types of Polyphenols
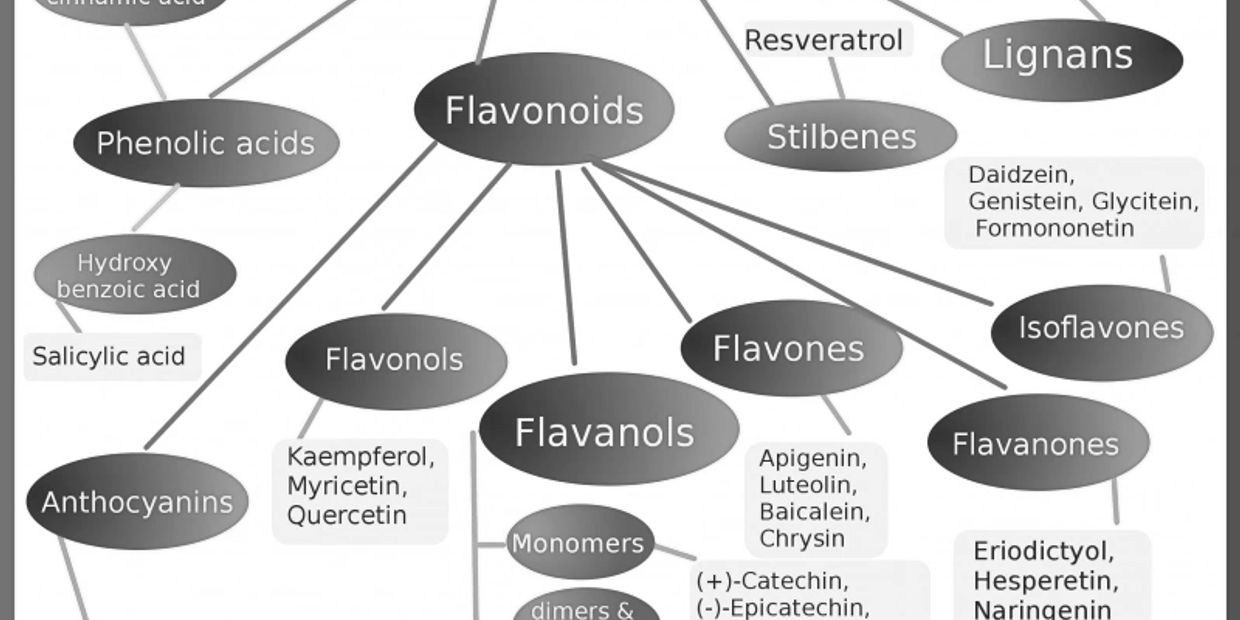
Furthermore, polyphenols can be partitioned into four groups, with additional subgroupings based on the number of phenol rings they contain and the structural elements connecting these rings together.
As a general guideline, foods contain complex polyphenol mixtures, with higher levels contained in the plant's external layers than the inner parts:
- Flavonoids, which both have antioxidant and anti-inflammatory characteristics, are encountered in fruits, vegetables, legumes, red wine, and green tea.
- ◦ Flavones
- ◦ Flavonols
- ◦ Flavanones
- ◦ Isoflavones
- ◦ Anthocyanidins
- ◦ Chalcones
- ◦ Catechins
2. Stilbenes, Found in red wine and peanuts (resveratrol is the most commonly known)
3. Lignans are mainly found in seeds such as flax, legumes, cereals, grains, fruits, algae, and certain vegetables.
4. Phenolic acids
- Hydroxybenzoic acids, found in tea
- Hydroxycinnamic acids are naturally found in cinnamon, but also in coffee, blueberries, kiwifruits, prunes, apples and cherries
Optimization of the nutritional content of phenols in diet
Flavonoids — compounds found in red wine and dark chocolate — are by far the most studied and known polyphenol class. Nevertheless, not even a single one polyphenol is necessarily better than the rest.
It is important to eat a wide variety of foods that are abundant in polyphenols in order to optimize the benefits of these natural health boosters. One of the best sources are high-quality seasonings, fresh fruits, vegetables and seeds. Nevertheless, bioavailability is also a question of how well the body can absorb the nutrients in the food One of polyphenols interesting facts is that they are fat-soluble.
in order for the organism to absorb them as quickly as possible they should be Included in the diet with fats
Eating a diet high in healthy fats will improve the body's ability to consume polyphenols. The way the plant is grown can also affect its capacity for healing. As noted in a paper ( American Journal of Clinical Nutrition May 2004: 79(5); 727-747 ) agricultural practices and industrial processes will decrease the health effects of polyphenols in the food.
Previous research ( Journal of Agricultural and Food Chemistry 2003: 51; 1237-1241) suggests that foods grown organically and in a sustainable manner yield statistically higher polyphenol levels compared to conventionally grown varieties. Freezing also retains a higher content of phenol than air-drying.
The Position of Polyphenols in Plants and Humans
Polyphenols give fruit, berries, and vegetables their vibrant colors and contribute to the bitterness, astringents, taste, aroma and oxidative stabilization of the foods.
They protect the plant against ultraviolet radiation, pathogens, oxidative damage and harsh climatic conditions.
Polyphenols have various biochemical and biological properties in the human body such as:
- Fight cancer cells and inhibit angiogenesis (growth of tumor-fed blood vessels)
- Ultraviolet radiation protection for skin
- Combat free radicals and reduce the onset of aging
- Promoting and maintaining brain health against dementia
- Inflammation management
- Support for healthy levels of blood sugar
- Protection of cardiovascular system
- Normal blood pressure regulation
Copyright © 2019 Louisiana Agriculture Preservation Society - All Rights Reserved.
Powered by GoDaddy Website Builder